|
10/10/2023 0 Comments Adenosine caffeine structure 
In scientific lingo, this process is called competitive inhibition because the caffeine is inhibiting the effects of adenosine by "competing" for its binding sites. To a nerve cell, the two compounds are virtually indistinguishable, and thus, will allow either caffeine or adenosine to bind to the adenosine-receptors. The key to caffeine's stimulating capabilities lies in its chemical structure, which is markedly similar to that of adenosine. Now let's bring caffeine into the equation. 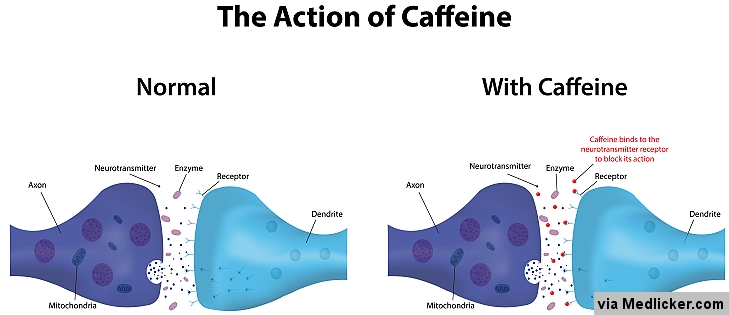
Adenosine binds to adenosine-receptors on neurons, facilitating the slow-down of your brain's signaling functions, and inducing a sensation we term "drowsiness." Adenosine concentration is highest in the brain when you have expended a lot of adenosine triphosphate (ATP),your body's energy currency,so it makes sense that you feel tired after working all day or running a marathon. When you feel sleepy it is because of molecules called adenosine floating around in your brain. Caffeine's "buzz," which is unregulated by governments worldwide, is responsible for the success of household commodities such as coffee, soda, energy drinks, and even certain pills.Ĭompetitive Inhibition: Caffeine's Modus Operandi When consumed by humans, the stimulant operates on the central nervous system (CNS) to produce an energizing effect that, among other things, wards off lethargy and promotes mental acuity. In its natural environment, it acts as an organic pesticide, protecting vegetation from insects with parasitic tendencies. Its widespread use, coupled with its lack of nutritional value, has attracted the condemnation of many dietary purists who brand caffeine as some sort of "demon compound." But to what extent is draining that café latte from Starbucks an exercise in sinfulness? While existing research offers conflicting opinions, the current consensus seems to be chanting "everything in moderation."Ĭaffeine is a chemical compound found in the beans, leaves, and fruits of over 60 known plants. According to a study conducted by New Scientist magazine, 90% of North American adults consume some form of caffeine on a daily basis, making this legal, psychoactive substance the world's most widely used drug. Whether it is a steaming mug of morning Joe or an afternoon pick-me-up soda, the world is addicted to caffeinated comforts. Let knowledge be the cure.Institution: Virginia Commonwealth University Powered by django-wiki, an open source application under the GPLv3 license. This article was last modified: April 20, 2015, 11:47 a.m. The functional groups of the caffeine are alkene, amide, and amine. The only difference between caffeine and theobromine is an additional methylene group in position 1 of caffeine. Both caffeine and theobromine also include an amide group, which is a functional group consisting of a carbonyl group bound to a C atom and a N atom, and an alkene, an unsaturated hydrocarbon containing at least one carbon-carbon double bond. Caffeine is built up of two carbonyl groups and 4 tertiary amines. 
Purines are heterocyclic aromatic compounds. These ring systems, named purines, are constructed from a pyrimidine (6 membered ring) and an imidazole ring (5 membered ring). The figure shows that caffeine is built up in two ring systems. Theobromine is called 3,7-dimethylxanthine, or 3,7-dimethylpurine-2,6-dione. Caffeine is called 1,3,7-trimethylxanthine, or 1,3,7-trimethylpurine-2,6-dione. Caffeine has a stronger stimulating effect on the CNS than theobromine.įigure: Chemical structure of caffeine and theobromine. Therefore, it may be difficult to separate them after analysis. Theobromine and caffeine are very similar, both physically and chemically. In plants which biosynthesize caffeine theobromine is also found. It is produced in plants such as coffee and tea from adenosine, which is found in DNA and RNA. Caffeine belongs to the group of substances called alkaloids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
